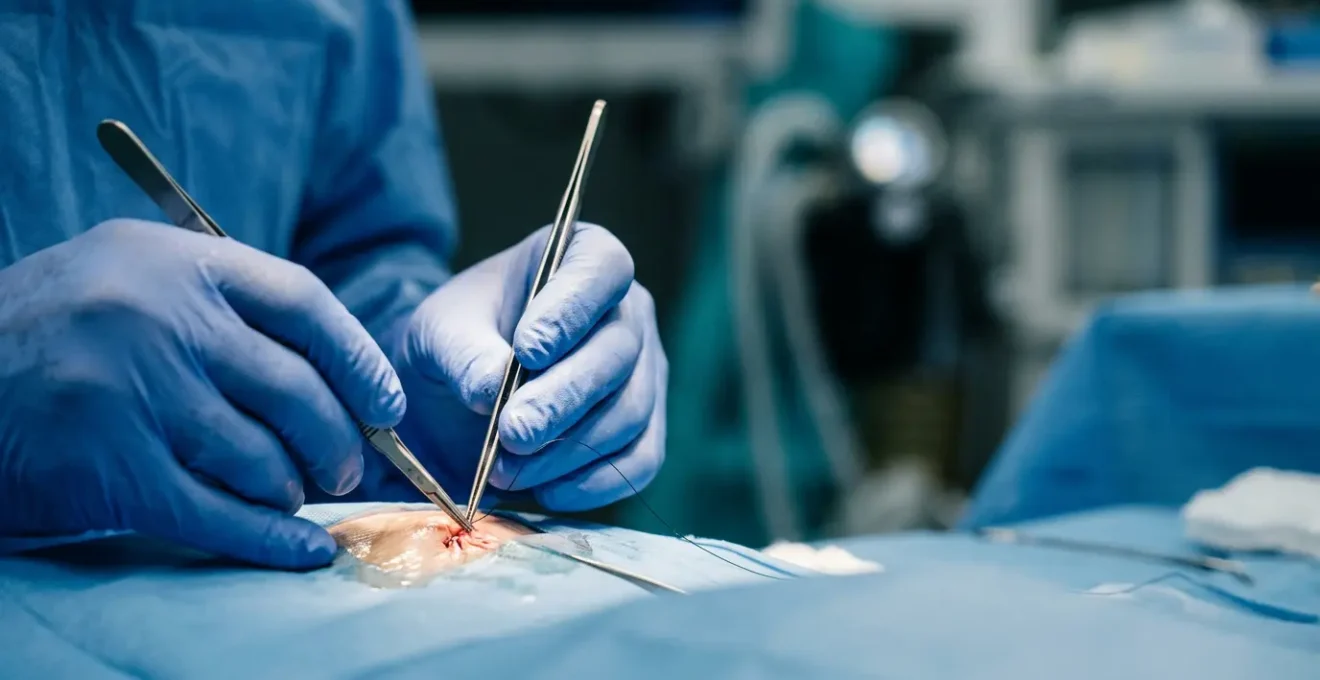
Reattaching a limb is not the final victory; it is the start of a biological marathon governed by unforgiving timelines and microscopic challenges.
- Success hinges on beating the 6-hour “warm ischemia” clock, the window before irreversible muscle death begins.
- Full sensory and motor recovery is a painstaking process, dictated by the slow, steady nerve regeneration rate of approximately 1 millimeter per day.
- Patient factors, such as smoking, can dramatically compromise healing and blood flow, even when the surgical procedure is technically perfect.
Recommendation: Understanding these fundamental biological limits is the first and most crucial step toward navigating a realistic and successful recovery journey.
The sudden, violent reality of a traumatic amputation is a moment of pure crisis. In the ensuing chaos, the immediate, overwhelming thought is one of restoration: can the severed part be put back? While the concept of reattaching a finger, hand, or arm might seem like a straightforward mechanical repair, the truth is far more complex and delicate. It is a world that transcends simple stitches and enters the realm of reconstructive microsurgery, where the battle for survival is fought on a scale measured in millimeters and microns.
Many assume that surgical skill is the only variable that matters. However, the success of a replantation is not just a testament to a surgeon’s steady hand; it is a race against the unyielding clock of cellular biology. The true challenges lie in the invisible details: the viability of tissues deprived of blood, the impossibly slow regrowth of severed nerves, and the microscopic integrity of newly formed blood vessels. This is where the miracle of microsurgery meets the cold, hard facts of human physiology. It is an intricate dance between surgical intervention and the body’s own capacity to heal.
This guide moves beyond the operating room to illuminate the critical, often hidden, factors that truly determine the outcome of a limb reattachment. We will dissect the timeline for tissue survival, decode the language of nerve regeneration, and uncover how a patient’s own choices can tip the scales toward success or failure. By understanding these principles, you gain the power to become an informed and active partner in your own extraordinary recovery.
Summary: How Microsurgery Can Save a Severed Limb When Standard Repair Is Not an Option
- Why You Only Have 6 Hours To Save A Detached Finger?
- How To Manage Expectations When Nerves Regrow At 1mm Per Day?
- The Vasoconstriction Effect: Why Smokers Fail Microsurgery 50% More Often?
- Muscle or Skin: Which Tissue Transfer Is Best For Covering Open Wounds?
- Why Doctors Sometimes Use Medical Leeches To Save Venous Congestion?
- Autologous Tissue or Implants: Which Offers More Natural Results?
- How To Care For The Biopsy Site To Avoid An Ugly Indentation?
- Reconstructive Interventions: When Is It Medically Necessary vs Cosmetic?
Why You Only Have 6 Hours To Save A Detached Finger?
The “golden hours” in trauma are not a myth; they are a biological mandate. For a detached body part, the primary enemy is ischemia: the cessation of blood flow. Without oxygen and nutrients, tissues begin to die. Muscle is the most vulnerable, with irreversible damage starting after just 6 hours of “warm ischemia” (at body temperature). This is the hard deadline that dictates the frantic race to the operating room. Research confirms the urgency, showing that the odds of survival for an early (0-6 hours) replantation are significantly higher than for a later one.
However, this clock can be slowed. Cooling the amputated part is the single most important action that can be taken at the scene. By properly preserving the digit—wrapping it in moist gauze, placing it in a sealed waterproof bag, and putting that bag on a mixture of ice and water—we induce “cold ischemia.” This dramatically lowers the tissue’s metabolic rate, acting as a biological pause button. It is a critical intervention that can extend the window of viability far beyond the standard six hours. A 2024 meta-analysis revealed that digits preserved in cold ischemia for over 12 hours had significantly higher odds of successful replantation than those with less than 12 hours of warm ischemia.
In the operating room, our first act is to restore this vital blood flow through vascular anastomosis, the microscopic connection of arteries and veins. Only once the part is “reperfused”—turning from a pale, lifeless white to a healthy pink—can we begin the meticulous work of repairing bones, tendons, and nerves. But that initial race against the ischemic clock determines if there is anything left to save at all.
How To Manage Expectations When Nerves Regrow At 1mm Per Day?
While restoring blood flow is the immediate miracle, restoring feeling and function is a painstaking marathon. Nerves are the body’s electrical wiring, and when severed, they do not simply reconnect. The distal part of the nerve (beyond the injury) dies off, and the proximal end must slowly, meticulously regrow its axon to find its target. This process occurs at a remarkably consistent, and often frustratingly slow, biological rate. As a reconstructive surgeon, I always tell my patients to anchor their expectations to this one number: 1 millimeter per day.
This is not an estimate; it is a well-established biological constant. Clinical evidence shows that at the average regeneration rate of 1 mm/day, a nerve injury at the wrist must regrow approximately 100 millimeters to reach the muscles and sensory receptors in the hand, requiring at least 100 days—over three months—before the first flickers of function can even begin. For an injury higher up the arm, the timeline can stretch to years. During this time, we track the nerve’s leading edge by tapping along its path, looking for the tell-tale tingling sensation known as Tinel’s sign, which marks the frontier of regeneration.
This slow journey is why sensory re-education is so critical. The brain must relearn how to interpret the new signals. The process involves methodical exercises, testing the recovering area’s ability to distinguish between sharp and dull, hot and cold, and different textures. It is a long, deliberate process of rebuilding a lost connection, one millimeter and one new sensation at a time.
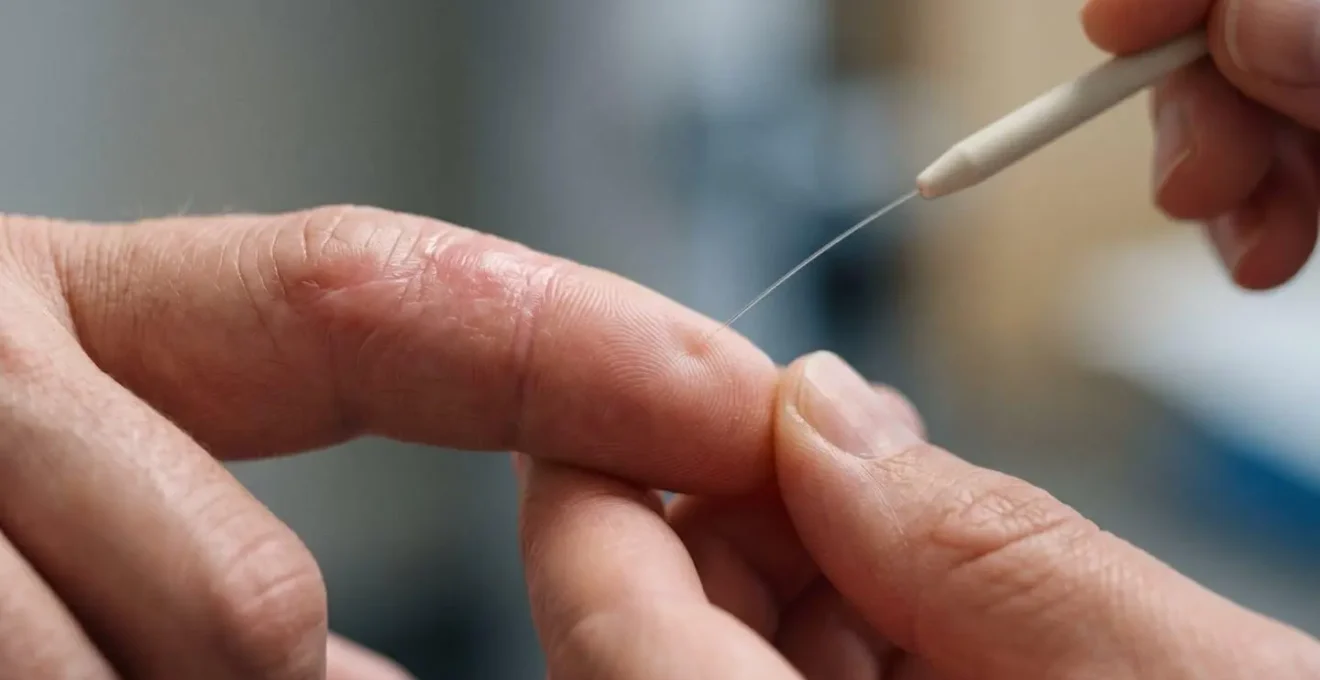
As you can see in this visualization of sensory testing, recovery is not a passive waiting game. It requires active participation to retrain the brain as the nerve endings slowly reawaken. Each small step of progress, measured in millimeters, is a victory on the long road back to function.
The Vasoconstriction Effect: Why Smokers Fail Microsurgery 50% More Often?
A perfectly executed microsurgical repair can be completely undone by a single factor within the patient’s control: smoking. Nicotine is a potent vasoconstrictor, meaning it causes blood vessels to clamp down, drastically reducing blood flow. For a newly reattached limb with fragile, microscopic repairs, this effect can be catastrophic. The new anastomoses—the sutured connections in vessels often less than 1mm in diameter—are exquisitely sensitive. The surge of nicotine from a single cigarette can cause these vessels to spasm and clot, starving the replanted tissue of the very oxygen it desperately needs.
While the “50% more often” figure is a common clinical aphorism, the hard data from scientific studies paints a detailed picture of the risks. It’s not just about the complete failure of the flap or replant; it’s about a cascade of healing complications that jeopardize the final outcome. Patients who smoke experience a battlefield of problems at the microscopic level, leading to visible, tangible consequences.
This is not a theoretical risk; it is a well-documented clinical reality. Surgeons will often delay or even refuse elective microsurgical procedures on active smokers until they have quit for a significant period. For trauma cases, the risk is accepted out of necessity, but the patient is warned in the strongest possible terms that their success is intrinsically linked to their ability to abstain from nicotine in any form.
Case Study: The Documented Impact of Smoking on Healing
A landmark retrospective study analyzed the outcomes of free-tissue transfers, a procedure similar to replantation. It compared 93 smokers to 51 nonsmokers. While the initial flap survival was surprisingly similar, a deeper look revealed the true cost. A study on the effects of smoking on 104 free-tissue transfers found that smokers suffered from significantly more delayed wound healing (35% vs. 24% in nonsmokers). More critically, they required additional surgical procedures to achieve final wound closure far more frequently (27% vs. 12%). This demonstrates that smoking creates a hostile environment for healing, turning a single surgery into a series of costly and painful interventions.
Muscle or Skin: Which Tissue Transfer Is Best For Covering Open Wounds?
When a traumatic injury results in a large open wound with exposed bone or tendons, simple closure is not possible. This is where the “reconstructive ladder” comes into play—a strategic framework surgeons use to select the appropriate tissue transfer. At the top of this ladder are free flaps: sections of a patient’s own tissue (skin, fat, muscle, and/or bone) that are detached from their original blood supply and reconnected microscopically to vessels at the recipient site. The choice of tissue is a critical strategic decision based on the specific needs of the wound.
A muscle flap is often the workhorse of reconstruction for deep, complex, or infected wounds. Muscle tissue has a robust blood supply, making it highly resistant to infection. It is also malleable, allowing it to fill “dead space”—the empty cavities left by the injury—and provide a healthy, vascularized bed for a skin graft. A fasciocutaneous flap, which consists of skin, fat, and the underlying fascia, is chosen when the primary goal is to provide stable, durable skin coverage in a single stage, such as over a joint or the sole of the foot. The decision is tailored: Muscle fights infection and fills volume; skin provides a simple, durable cover.
Regardless of the tissue type selected, the technical execution remains the pinnacle of surgical precision. These are complex, lengthy operations that depend on the surgeon’s ability to perform flawless vascular anastomoses under a microscope. When undertaken by a specialist, the results are consistently reliable. In fact, expert microsurgical centers achieve replant and free flap viability rates of at least 80%, a testament to the power and predictability of these advanced techniques. The art lies not just in the execution, but in choosing the right tool for the job.
Why Doctors Sometimes Use Medical Leeches To Save Venous Congestion?
One of the most feared complications after a successful replantation is venous congestion. While the arterial inflow (the “in” pipe) might be working perfectly, the venous outflow (the “out” pipe) can become blocked. Veins are more fragile and lower-pressure than arteries, making them prone to kinking or clotting. This creates a microscopic traffic jam. Blood flows into the replanted part but cannot get out, causing it to become swollen, purple, and engorged. If not resolved, this pressure will choke off the arterial supply, leading to tissue death.
When surgical revision is not possible or has failed, we turn to one of nature’s most brilliant, albeit unsettling, solutions: medicinal leeches (Hirudo medicinalis). This is not an archaic practice; it is a highly effective, modern technique for flap salvage. The power of the leech lies in its saliva, which contains a potent cocktail of bioactive compounds.
Leeches produce an enzyme in their saliva called hirudin. Hirudin is a powerful anticoagulant.
– University of Iowa Carver College of Medicine
This natural anticoagulant is the key. As the leech draws blood, the hirudin prevents clotting in the microvasculature, allowing the congested venous blood to escape. The leech acts as a temporary, living drainage system, decompressing the tissue and giving the body time to form new, natural venous outflow channels. The effect is often immediate and dramatic.

The success of this intervention is well-documented. While it may not save every compromised flap, it provides a crucial lifeline. A study of flap salvage outcomes revealed that of those treated with leech therapy for venous congestion, 33.3% were completely salvaged, and another third were partially salvaged. In the world of microsurgery, where every percentage point matters, the humble leech remains an indispensable tool for turning a near-certain failure into a remarkable success.
Autologous Tissue or Implants: Which Offers More Natural Results?
When reconstructing a defect, surgeons face a fundamental choice: use the patient’s own tissue (autologous reconstruction) or use a synthetic material (alloplastic reconstruction, i.e., implants). For achieving a truly “natural” result—one that feels, ages, and heals like the body’s own—autologous tissue is almost always the superior choice. Using a patient’s own skin, fat, or muscle means the body recognizes the tissue, readily integrates it, and establishes a permanent blood supply. It becomes a living, dynamic part of the reconstruction that will change and adapt with the patient over their lifetime.
Implants, while useful for providing structure (like in breast or craniofacial reconstruction), are always foreign bodies. They carry risks of infection, rejection, and capsular contracture (the formation of hard scar tissue around the implant). They do not heal or age naturally and may require replacement over time. Autologous tissue, by contrast, is “you.” A flap of skin and fat taken from the abdomen to reconstruct a breast, for example, will feel soft, warm, and natural in a way no silicone implant ever can.
However, “natural” can have two meanings: appearance and function. While autologous tissue provides the most natural look and feel, achieving natural function, especially after nerve injury, remains a formidable challenge. Even with perfect microsurgical nerve repair using autologous nerve grafts (harvesting a less important sensory nerve to bridge a critical motor or sensory gap), the functional outcome is never guaranteed. The sobering reality is that less than half of patients who undergo nerve repair after injury regain good to excellent motor or sensory function. The connection is made, but the quality of the signal that gets through is often imperfect.
How To Care For The Biopsy Site To Avoid An Ugly Indentation?
In the focus on saving a limb or reconstructing a major defect, it is easy to overlook the site from which the tissue was taken: the donor site. Harvesting a free flap is essentially creating a second, planned surgical wound. The primary goal of managing this site is not just to close it, but to do so while minimizing cosmetic deformity and functional loss. One of the most common patient complaints after free flap surgery is an unsightly depression, indentation, or a poorly healed scar at the donor site.
A proactive and meticulous post-operative care plan is essential to prevent these issues and ensure an aesthetic outcome. This is not a passive process; it requires a dedicated regimen of therapies designed to control swelling, manage scar formation, and restore contour. Neglecting the donor site can lead to long-term dissatisfaction, even if the reconstruction itself is a success. The goal is a “win-win”: a successful reconstruction and a donor site that the patient can live with comfortably.
The following steps represent a comprehensive strategy for optimizing the healing and final appearance of the donor site. Adherence to this protocol can make the difference between a barely noticeable scar and a permanent, disfiguring indentation.
Action Plan: Your Donor Site Care Protocol
- Immediate Compression: Apply compression garments immediately post-operatively to reduce swelling (edema) and help shape the contour of the donor site.
- Meticulous Scar Massage: Once the wound is sufficiently healed, initiate regular scar massage protocols to improve the scar’s pliability and prevent it from becoming hard and contracted.
- Silicone Sheeting Application: Utilize medical-grade silicone sheeting for 12-24 hours daily. This helps to flatten and soften raised scars by hydrating the tissue and applying gentle pressure.
- Consider Secondary Fat Grafting: For any persistent contour depressions or indentations, secondary procedures like fat grafting can be considered to restore lost volume and improve the aesthetic appearance.
- Monitor and Rehabilitate Function: For donor sites where muscle was harvested (e.g., the latissimus dorsi), monitor for any weakness and coordinate with physical therapy to rehabilitate and strengthen the remaining muscles.
Key Takeaways
- Cold preservation is paramount for extending the critical 6-hour survival window for a detached part by slowing cellular metabolism.
- Nerve recovery is a long, predictable marathon, not a sprint, governed by the biological constant of approximately 1 millimeter of growth per day.
- Patient health choices, especially abstaining from nicotine, are as critical as surgical skill for ensuring the survival and optimal healing of microscopic vessel repairs.
Reconstructive Interventions: When Is It Medically Necessary vs Cosmetic?
The line between reconstructive and cosmetic surgery can sometimes seem blurry, but it is defined by a clear and fundamental principle: function. Cosmetic surgery aims to enhance a normal appearance, while reconstructive surgery aims to restore form and, most importantly, function lost due to trauma, disease, or congenital defect. In the world of microsurgery, this distinction is often dictated by strict biological timelines that represent a point of no return.
A perfect example is the reinnervation of a muscle. When a nerve that controls a muscle is severed, the muscle is paralyzed. It can be saved, but only if its nerve supply is restored within a specific window of time. After that window closes, the muscle’s motor end plates—the specialized junctions where nerve and muscle communicate—begin to degenerate irreversibly. No amount of surgery can bring that muscle back to life once it has crossed this threshold. The intervention is, therefore, not a matter of choice or aesthetics; it is a medical necessity to prevent permanent functional loss.
This principle is absolute. There is an accepted window of 12-18 months for muscle reinnervation to occur before irreversible degeneration makes functional recovery impossible. A surgeon performing a nerve transfer at 6 months is restoring function; a surgeon asked to do the same procedure at 24 months is attempting the impossible. The goal of reconstruction is to rebuild what was lost—a breast after a mastectomy, a thumb’s ability to pinch, a jaw’s ability to chew, a leg’s ability to bear weight. When function is on the line, the intervention is always a necessity.
Armed with this knowledge of what is possible and the biological laws that govern it, the most critical next step is an open and detailed conversation with your reconstructive team. This will allow you to align expectations, understand your unique situation, and co-create the most effective path toward your recovery.